
Molecular tumor profile in FFPE tissue sample and liquid biopsy (prime DX® & prime DX® Liquid)
Genekor's prime DX® Combo enables the simultaneous testing of FFPE tissue and liquid biopsy samples, providing a comprehensive molecular profile by exploiting the advantages of both procedures.
Why Choose Simultaneous Tissue and Liquid Biopsy Testing?By integrating both approaches, clinicians gain a more comprehensive understanding of a patient’s tumor biology resulting in better-informed treatment decisions. Concurrent testing can lead to:
State-of-the-Art Testing Platforms: Utilizing advanced Next-Generation Sequencing (NGS) technologies, in combination with
commercially available and proprietary bioinformatics tools, our integrated testing approach ensures accurate, reliable, and comprehensive data.
Experienced Scientific Team: Our team consists of specialized molecular biologists and geneticists who translate genomic data into clinically actionable insights, supporting and optimizing personalized therapeutic approaches.
Two Perspectives, One Powerful Profile: Combining tissue and liquid biopsy for precision care.
The prime DX® assay is performed in a CAP-accredited laboratory, while the analysis of selected genes and biomarkers falls within the scope of accreditation according to ISO 15189:2022.
Next-Generation Sequencing (NGS) is performed using the MGI DNBSEQ-T7 and MGI DNBSEQ-G400 platforms, which, along with the reagents used, are CE-IVD, ensuring high sensitivity and specificity in multi-sample processing.
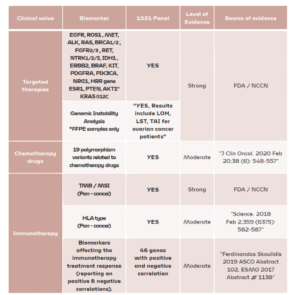
A multi-gene panel generates a large volume of multidimensional data, supporting personalized treatment strategies for patients.
This approach increases the likelihood of identifying appropriate therapeutic targets, whether within approved indications
(on-label), beyond them (off-label), or for potential inclusion in clinical trials.
At the same time, it requires minimal tissue or liquid biopsy sample input, while enabling the simultaneous analysis of multiple genes in a faster and more cost-efficient manner.
Genekor ensures a high level of quality, reliability, and security through internationally recognized accreditations and certifications.
*Within the official scope of accreditation.
Which patients are eligible for the test?
All patients with a solid tumor are eligible.
How long does it take to get my results?
15 business days
What kind of sample and how much is required for the test?
For prime DX® Combo analysis, we need a paraffin block containing the tumour tissue. We need one paraffin block or four 3 µm paraffin sections: in positive charged slides (air dried at room temperature, not baked), placed on top of the slide, without folds and six unstained slides of 10μm.
Also, we need blood in special vials that you will receive from Genekor. (Cell-Free DNA BCT®(10ml)
Why is it necessary to analyse so many genes?
A multi-gene panel results in the generation of a large amount of multi-level information of useful value greatly increasing the likelihood of finding a personalised cancer treatment plan for the patient.
Also, it has been shown that the more genes analysed, the more reliable the results for TMB.
Are there any special transportation conditions?
The blood sample (liquid biopsy) should be transported in conditions up to 20°C.
Why should I sign the consent form?
For the use of the patient’s genetic material for testing, ELOT EN ISO9001:2008 and ELOT EN ISO15189:2012 require the written consent of each patient.
Does any public/private insurance cover the test?
For information about the cost coverage of the test you should contact your insurance company.
How can I pay for the test?
Payment can be made by bank transfer or debit/credit card.
How can I send my sample?
To schedule pickup and return of your sample, please contact us.
How will I receive my results?
Your results are shared with your GP via email.
Our Customer Service Team is committed to answering your questions about the services offered by Genekor. If you are interested in any of the tests offered by Genekor, please contact us directly.
*To complete the test, you are required to complete and send the Consent form that you will find on the link below.
*For more information on scientific content please contact: scientific.support@genekor.com
*Download the promotional brochure here.